Cryogenic Technology: Principles, Systems, and Industrial Applications

Cryogenic technology integrates physics, engineering, and industrial design in ways that do a lot more than merely make things colder. It's about making regulated areas where gases become liquids, biological materials stay the same, and materials reveal features that aren't evident at normal temperatures. These conditions make some processes possible that wouldn’t be possible in any other way for industries like pharmaceuticals and semiconductor manufacturing.
This article explains the basic ideas behind cryogenic technology — what temperature ranges are considered cryogenic, how those very low temperatures are reached, which cryogenic systems and gases are used, what safety risks need to be managed, and where industrial cryogenics really adds value. It also looks at why standard cryogenic refrigeration stops working long before it reaches the lowest temperatures, and how new engineering methods are changing the way cryogenic systems are built.
What Is Cryogenic Technology?
Cryogenic technology is the science and engineering field that deals with making, keeping, and working with very low temperatures, usually anything below −160 °C (around 113 K) in most factories. The word itself comes from the Greek roots kryos, meaning frost, and genics, meaning to produce. The International Congress of Refrigeration in Washington, D.C. set 120 K (−153 °C) as the dividing line between standard refrigeration and conventional cryogenic refrigeration in 1971.
Cryogenic Temperature Ranges Explained
Cryogenic temperatures cover a wide range, with each level defined by the cryogenic gases that turn into liquids there. Methane and LNG liquefy at about −161 °C. Oxygen boils at −183 °C. It is used in medicine, to make steel, and as rocket fuel. At −186 °C, argon is next. This is a common temperature for making processing environments that don't react. Nitrogen is one of the most common cryogenic fluids. It boils at −196 °C and can be used for anything from freezing food to covering things in an inert blanket. At −253 °C, hydrogen changes from a gas to a liquid. This is useful for rocket propulsion and fuel cells. Helium, the coldest of the group, boils at −269 °C and is essential for MRI magnets, superconducting systems, and particle physics.
Most industrial cryogenic refrigeration systems work in the higher part of this range, which is about −50 °C to −160 °C. . When temperatures fall below −196 °C, engineering complexity escalates significantly, requiring cryogenic gases like helium and hydrogen.
How To Get Cryogenic Temperatures
There are different ways to get to cryogenic temperatures, and the best one depends on the temperature you want to reach, the amount of cooling power you need, and the environment. Most factories use more than one method in a single cryogenic system.
Thermal Conduction Cooling
Thermal conduction cooling uses cryogenic liquids, like liquid nitrogen, as a direct coolant. It moves them around by touching them directly, which cools them down. It can be easily made bigger, but it needs a constant supply of cryogenic liquid to keep going.
Evaporative Cooling
Evaporative cooling works by letting a cryogenic liquid boil, which cools the air around it. Liquid nitrogen that evaporates at −196 °C is a popular choice for food freezing tunnels, cryotherapy chambers, and biological storage facilities because it cools things down quickly and doesn't require a lot of complicated machinery.
Joule–Thomson Expansion
The Joule–Thomson effect occurs when a real gas is pushed through a restriction from high pressure to low pressure. Below the gas’s inversion temperature, this expansion causes a noticeable drop in temperature. This principle is used to liquefy hydrogen and helium and serves as the basis for the Linde–Hampson liquefaction cycle.
Adiabatic Demagnetization
Adiabatic demagnetization refrigeration (ADR) takes over when temperatures drop below 1 K, where mechanical and gas-based cooling methods can no longer keep up. It uses the magnetocaloric effect — magnetizing and then demagnetizing a paramagnetic material in controlled conditions to rapidly drop its temperature. This method is used in space instruments, physics research labs, and the growing field of quantum computing research.
Cryogenic Systems and Infrastructure
A cryogenic system is a fully integrated infrastructure in which every part is designed to work reliably at temperatures where normal materials break down, lubricants freeze solid, and even small mistakes in thermal management can have big consequences.
Main Components of a Cryogenic System
Industrial cryogenic systems include several key components. The cryogenic chiller or cryocooler is the main cooling unit, responsible for reaching and holding target temperatures.
Some of them are air-cycle, Stirling, Gifford-McMahon, pulse tube and mixed-refrigerant designs:
- Vacuum-jacketed Transfer lines with vacuum jackets move cryogenic fluids while keeping heat out. Dewars and storage vessels are double-walled, vacuum-insulated containers that hold cryogenic liquids such liquid nitrogen, helium, and hydrogen.
- Cryogenic pumps carry the liquids from storage to processing equipment. Air separation and liquefaction need heat exchangers that work at efficacy levels above 95%.
- Control systems connect everything and keep an eye on temperature and pressure automatically. They also set off safety measures when they are needed.
Why Vacuum Insulation Is Important
Vacuum insulation is highly important since even a little bit of heat from outside can have a large effect at very low temperatures. There is a space between two walls that are very near to each other. This stops both conductive and convective heat transfer, so only radiative transfer is left to cope with.
Cryogenic Gases and What They Are Like
You can't use the same cryogenic gases in industrial settings. Each one has a different boiling point, density, reactivity profile, and cost structure that decides where and how it is used. To design cryogenic systems, you need to know these properties.
Nitrogen, Oxygen, and Argon
Nitrogen (N₂) is the most common cryogenic gas in the world. It boils at −196 °C. It has no smell, no color, is not flammable, and is chemically inert, which is why it is the best choice for freezing food, cryopreservation, inert blanketing in manufacturing, and cooling research.
Oxygen (O₂) boils at −183 °C and is a strong oxidizing agent used in making steel, launching rockets, and medical gas systems.
Argon (Ar) boils at −186 °C and doesn't react with other chemicals, even when it's hot.
Helium and Hydrogen
At −269 °C, helium (He) has the lowest boiling point of any element. In addition, it is the only coolant that works for superconducting magnets in particle accelerators, MRI machines, and NMR spectrometers. Because helium is a limited resource that can't be replaced, and its price has gone up a lot in the last few decades, systems that use it must be built from the ground up with the goal of minimizing loss.
Hydrogen (H₂) boils at −253 °C and is becoming more important to store energy for rocket propulsion, fuel cell technology, and fusion research. Because it can catch fire in air from 4% to 75%, safety engineering is needed at all levels, and cryogenic system design must take into account how hydrogen makes metals brittle.
Safety and Risks in Cryogenic Engineering
Injuries from Extreme Cold
If you touch cryogenic liquids or their vapors directly, they can freeze human tissue almost instantly, causing frostbite, cold burns, and damage to deep tissue. Face shields, cryogenic gloves, and insulated clothing are all required when working with cryogenic liquids.
Risks of Fire, Explosion, and Pressure
Cryogenic systems that use flammable gases like hydrogen and LNG need special engineering controls to keep fire and explosions from happening. Liquid oxygen can't catch fire on its own, but it speeds up the burning of things that wouldn't normally catch fire. The Boiling Liquid Expanding Vapor Explosion (BLEVE) is a very dangerous pressure hazard. When a pressurized vessel breaks, the liquid flash-boils almost instantly, and the vapor expansion sends out a shockwave that can destroy things.
This risk applies to non-flammable cryogens like liquid nitrogen as well as flammable gases. Reliable pressure relief systems, regular vessel inspections, and strict storage conditions are the primary engineering defenses.
Rapid gas expansion is another serious risk. One liter of liquid nitrogen expands to approximately 700 liters of gas at room temperature, displacing oxygen and creating life-threatening asphyxiation conditions with no warning.
Loss of Valuable Gases
Helium loss is also a significant operational and financial challenge. Because helium is extracted from natural gas wells and cannot be recovered once it escapes into the atmosphere, boil-off losses translate directly into major costs. Facilities running superconducting systems typically invest heavily in helium recirculation and recovery systems and reducing or eliminating helium dependence has become an increasingly important design criterion.
Industrial Applications of Cryogenic Technology
In medicine and pharmaceuticals, cryopreservation of blood plasma, stem cells, embryos, and tissue samples depends entirely on consistent temperature maintenance — any thermal excursion can render biological materials completely unusable.
Uses in medicine and pharmacy
Certain vaccines, including mRNA-based products, must be stored between −60 °C and −90 °C to remain viable. Pharmaceutical freeze-drying (lyophilization) relies on cryogenic refrigeration to remove moisture while preserving biological structure and bioactivity.
Industry of Food and Drink
In the food and beverage industry, rapid freezing with liquid nitrogen produces smaller ice crystals, causing significantly less cellular damage and better preservation of texture, flavor, color, and nutritional content compared to slower mechanical freezing. Cryogenic systems also underpin cold chain integrity across international distribution networks for high-value perishables.
LNG, Energy, and Hydrogen
In energy, LNG — natural gas liquefied at around −161 °C to −162 °C — shrinks to roughly 1/600th of its original volume, making large-scale sea transport and storage genuinely practical. Liquid hydrogen at −253 °C enables the high-density storage needed for fuel cell vehicles, aircraft, and industrial decarbonization processes.
In semiconductor manufacturing, cryogenic etching methods like Deep Reactive Ion Etching (DRIE) enable precise material removal without damaging surrounding structures.
Electronics and Semiconductors
Cryogenic cooling also stops oxidation during the most delicate steps of making something. Superconductivity is when there is no electrical resistance at all below a certain temperature. This makes it possible to build powerful electromagnets for MRI machines, particle accelerators, and quantum computers. Quantum processors today work close to absolute zero, which means that cryogenic technology is not just a tool for quantum computing; it is the basis for the whole field.
Scientific Research and Particle Physics
The Large Hadron Collider at CERN is at the cutting edge of basic physics. It uses superfluid helium to cool about 27 kilometers of superconducting magnets to 1.9 K colder than outer space. This creates the magnetic fields needed for high-energy particle experiments.
Cryogenic Engineering vs. Conventional Refrigeration
Conventional vapor-compression refrigeration works well down to about −40 °C to −50 °C. Below that, standard refrigerants including HFCs and HFOs can no longer function effectively as heat transfer media.
Limitations of Refrigerants at Ultra-Low Temperatures
Cascade refrigeration systems can push the range further by chaining multiple refrigerant circuits together, but at the cost of added complexity, multiple failure points, and poor energy efficiency at partial loads. Below −50 °C, conventional compressor oils thicken dramatically, increasing mechanical resistance and eventually causing system failure, making oil-free compressor technology a requirement for reliable cryogenic operation.
Regulatory pressure adds further complexity. The EU F-Gas Regulation and equivalent frameworks elsewhere are accelerating the phase-out of high global warming potential synthetic refrigerants, creating real compliance uncertainty for HFC-based systems over their operational lifetimes.
Air-Cycle and Alternative Approaches
Air-cycle refrigeration uses a closed thermodynamic cycle to cool things down by compressing, exchanging heat, and expanding air through a turbine. Air doesn't change phases within the operational range, and air bearings don't need lubricating oil. This means that air-cycle systems don't have any of the refrigerant regulatory or supply-chain risks that synthetic alternatives do. They don't cause any global warming, don't need to be refilled with refrigerant, and aren't subject to F-Gas or EPA rules. This means they will be structurally safe as more rules are put in place for synthetic refrigerants.
.png)
How Mirai Intex Fits into Cryogenic Cooling Architectures
Mirai Intex is a developer of air-cycle refrigeration technology, not a cryogenic gas supplier. Its role within the cryogenic systems ecosystem is as a provider of cooling infrastructure that replaces or works alongside gas-based cryogenic refrigeration systems, particularly in the temperature range where conventional refrigeration runs out of road.
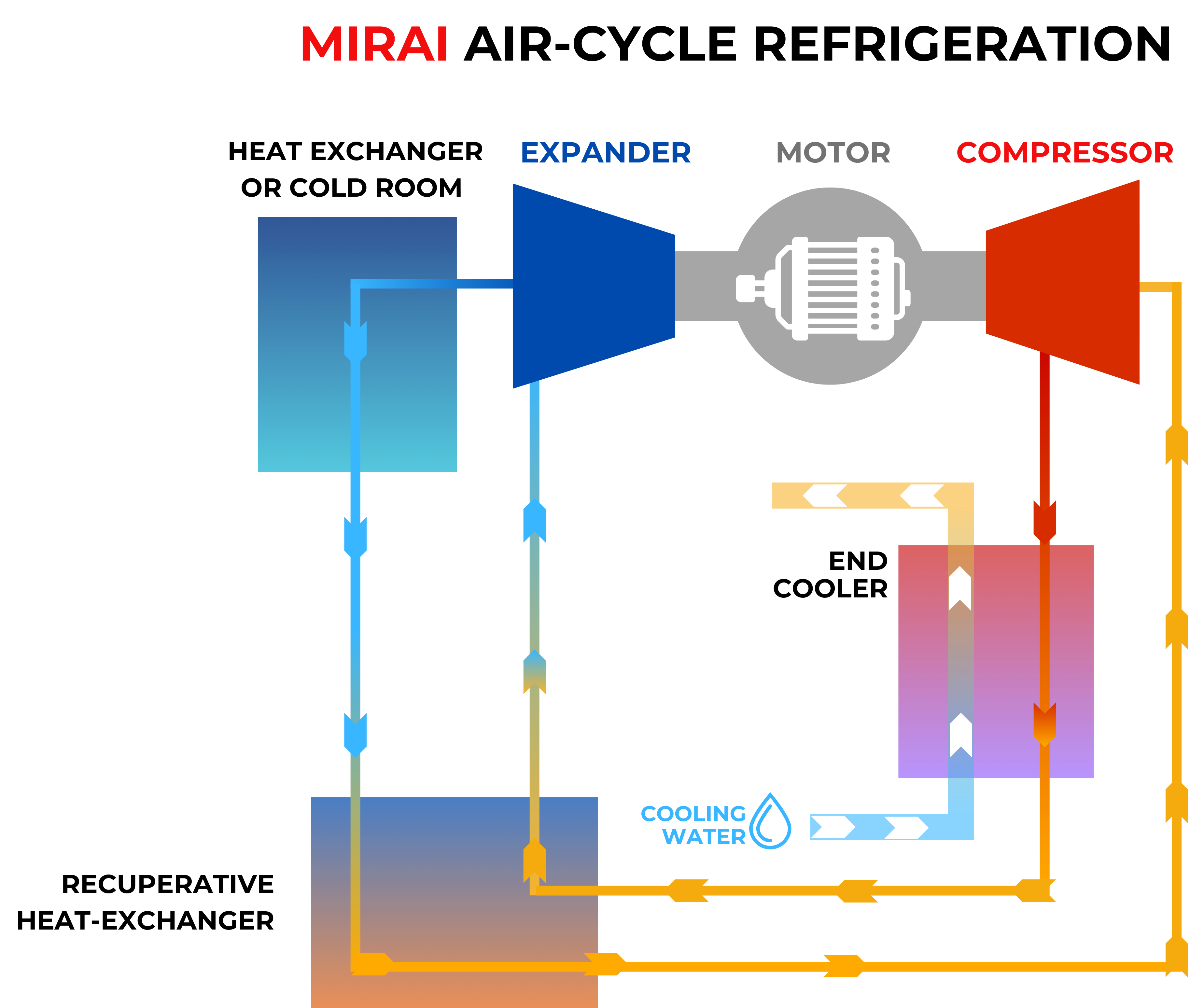
Air-Cycle Refrigeration for Cryogenic Ranges
The MIRAI X CRYO system operates from +90 °C down to −160 °C, using air as the sole refrigerant in a closed cycle. That range covers the full upper cryogenic range.
The ULT Liquid Cooler operates on the same air-cycle principle, with temperature range from -40 °C down to −160 °C, with both cooling capacity and temperature regulated by turbo module speed, enabling precise, continuous control without stop-start cycling — a meaningful reliability advantage for pharmaceutical storage, semiconductor process cooling, and cryotherapy applications.
.png)
Benefits for Industrial and Medical Systems
By utilizing a closed-loop air cycle, our system removes the burden of boil-off losses and helium market instability. We have engineered out the risks of process contamination and ultra-low temperature failure modes by leveraging oil-free air bearings.
The result is a highly responsive cooling solution that scales energy consumption to match your actual load, outperforming traditional constant-power cascade systems. With no need for refrigerant refills or specialized maintenance, your day-to-day operations become significantly simpler and more reliable.
Conclusion
Cryogenic technology is a foundational discipline for a growing number of industries — from pharmaceutical cold chains and semiconductor fabrication to LNG infrastructure and scientific research. As demand expands, so does the engineering complexity, with cryogenic systems required to simultaneously manage extreme temperatures, high-value gases, regulatory requirements, and serious safety risks.
The architecture of cryogenic refrigeration is shifting. Where cascade systems and cryogenic gas consumption were once the only practical options below −50 °C, air-cycle refrigeration now offers a genuinely viable alternative across a substantial portion of the industrial cryogenic range — without synthetic refrigerants, without helium dependence, and without the regulatory exposure that is increasingly weighing on conventional systems.
For engineers and procurement teams evaluating cryogenic cooling infrastructure, the right question is not simply which technology can reach the required temperature — it is which technology does so with the reliability, regulatory stability, and total cost of ownership that long-term industrial cryogenics operations demand.
If your application requires cooling in the −40 °C to −160 °C range and you are assessing your system architecture, contact the Mirai Intex technical team for a process-specific discussion.





